Join Our Newsletter And Save 50%* On Your First Order
Get the lowdown on exciting updates, new products, exclusive discounts, and more.
*Good on WellspringCBD and Weltaday branded products only.
Get the lowdown on exciting updates, new products, exclusive discounts, and more.
*Good on WellspringCBD and Weltaday branded products only.
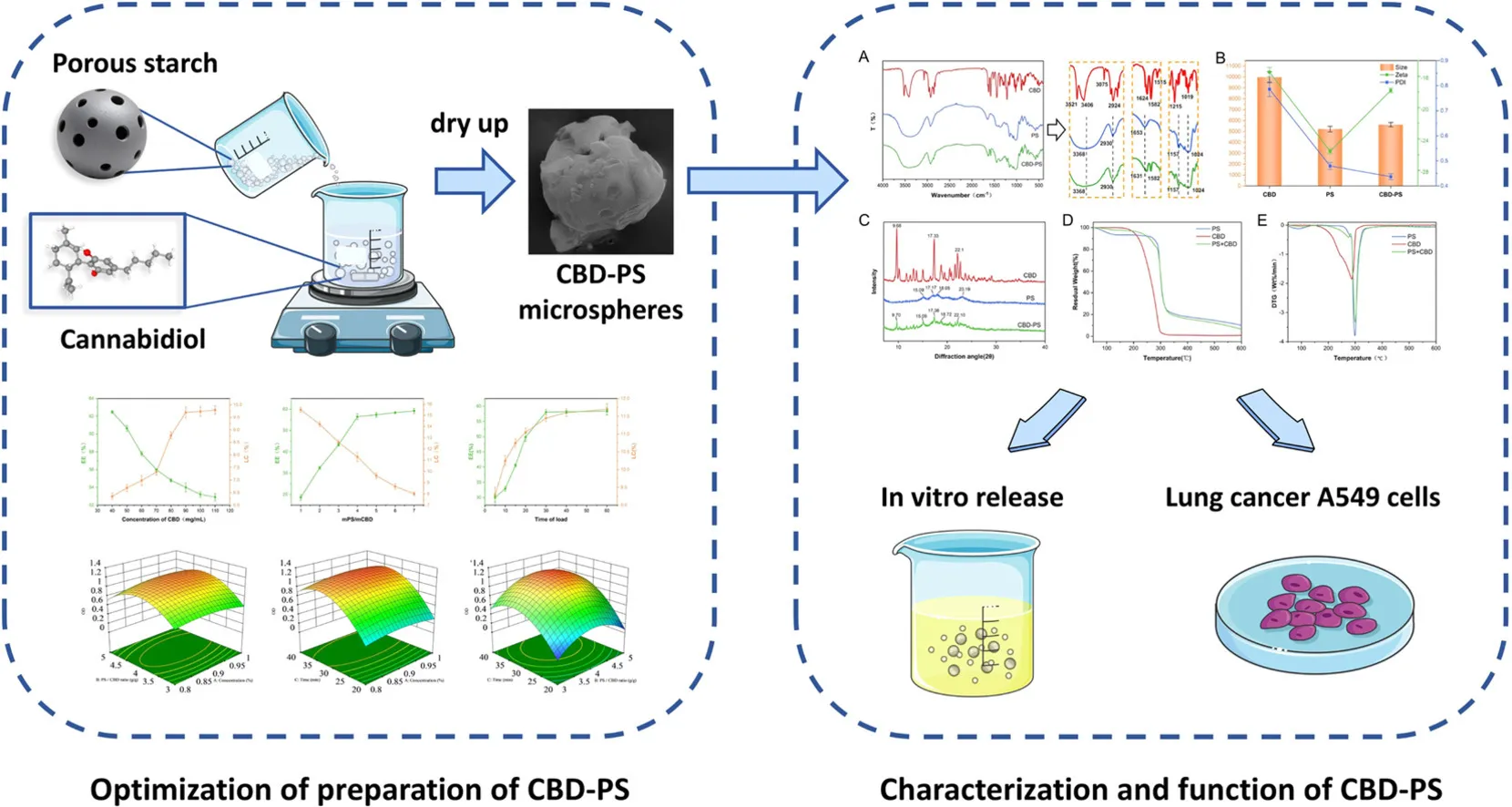
Recent research published in the Journal of Future Foods reveals a transformative advancement in cannabidiol (CBD) delivery technology that addresses one of the compound’s most persistent limitations: poor water solubility. Researchers have developed CBD-loaded microspheres using food-grade porous starch, a biocompatible material engineered to dramatically enhance both stability and absorption. The results are striking—the new formulation achieves a 16-fold improvement in water solubility compared to conventional CBD products, fundamentally changing how the body processes and utilizes this therapeutic compound.
This innovation directly tackles what has historically limited CBD’s clinical efficacy and commercial viability. As the expert analysis notes, “Advanced starch-based delivery systems represent a transformative solution to CBD’s fundamental bioavailability challenge by dramatically enhancing water solubility (16-fold improvement) and accelerating gastrointestinal absorption rates (2.6-4.4x increase).” The practical implications are substantial:
Gastrointestinal Release Performance:
-Gastric conditions: 2.6x faster release rates
-Intestinal absorption: 4.4x increased bioavailability
-Formulation stability: CBD stabilized in amorphous form within starch matrix
Beyond absorption metrics, this research provides compelling mechanistic evidence that positions CBD as a candidate for evidence-based medical-grade therapeutics. When tested on A549 lung cancer cells, the CBD-starch formulation demonstrated over 52% inhibition of cancer cell growth at 50 micrograms per milliliter—a result linked to specific biological pathways rather than non-specific toxicity.
The research identified two critical mechanisms driving this anti-cancer activity:
This mechanistic validation is crucial. As the clinical segment analysis emphasizes, “The mechanistic validation through mitochondrial disruption and PPARG gene modulation in lung cancer models establishes a scientific foundation for moving CBD from nutraceutical positioning toward evidence-based medical-grade therapeutics.” This distinction matters significantly for regulatory pathways and clinical credibility.
The enhanced understanding of this research creates opportunities across multiple audience segments. For consumers seeking genuine wellness solutions, the starch-based delivery system makes CBD therapeutically accessible by solving a fundamental absorption problem—lower doses could deliver better results at potentially lower costs. This addresses a critical barrier: affordability.
The food-grade starch carrier also democratizes CBD access by enabling integration into mainstream food products rather than limiting distribution to specialty supplement channels. This expansion pathway is particularly significant for:
While these findings are genuinely promising, maintaining scientific integrity requires acknowledging substantial gaps between laboratory evidence and real-world application. The expert consensus emphasizes: “These findings remain preliminary and confined to controlled in vitro conditions. Significant gaps remain between laboratory efficacy and real-world human applications, including questions about dosage optimization, long-term safety, and reproducibility in clinical settings.”
Key limitations to consider:
This measured perspective actually strengthens credibility by distinguishing between validated laboratory science and speculative clinical claims—a critical distinction for building trust with informed audiences.
This starch-based delivery system represents a genuine breakthrough in CBD therapeutics—not through marketing hyperbole, but through measurable improvements in bioavailability and demonstrated anti-cancer mechanisms. The 16-fold enhancement in water solubility and 4.4-fold increase in intestinal absorption address real pharmacokinetic barriers that have historically limited CBD’s therapeutic potential.
However, the journey from laboratory promise to clinical reality requires sustained investment in human studies, regulatory navigation, and manufacturing scale-up. Organizations that combine this research foundation with rigorous clinical validation will be positioned to lead the transition of CBD from nutraceutical commodity to evidence-based therapeutic—ultimately serving consumers seeking genuine health benefits backed by science rather than marketing claims.